GLP-1 adoption has already influenced consumption patterns, leading to a 5.3% drop in grocery spending within six months. Consumers are cutting back on snacks, sweets, and baked goods while moving toward nutrient-dense options such as yogurt, fruit, nutrition bars, and meat snacks.
It is a structural change in food demand where smaller, more functional portions are becoming the new norm. That’s why food companies are designing foods and ingredients that support endogenous GLP-1 release while maintaining protein, fiber, and micronutrient intake, and digestive comfort.
Nestlé’s Vital Pursuit launch and subsequent protein-technology work for GLP-1 users and weight management show that large food companies now see this as a distinct product platform rather than a short-term marketing theme. That distinction matters for R&D, because it shapes what can be claimed, what can be formulated, and what can scale.
This analysis maps the current research landscape of GLP-1 in functional foods, where the science is credible, which ingredient systems are most active, where companies are placing bets, and where the white space remains.
Why GLP-1 Is Becoming Critical in Functional Foods
The technical gap in current weight-management foods lies in nutrient density per bite, satiety per serving, and glycemic control without poor texture or taste. GLP-1 sits inside all three.
Most foods that claim satiety still rely on broad signals such as protein content, fiber load, or low glycemic response. Those features matter, but they don’t always produce a strong or reproducible hormonal response. GLP-1 matters because it links food intake to appetite control, gastric emptying, insulin release, and postprandial glucose handling.
Many ingredients that trigger GLP-1 release are bitter, and scaling these solutions while maintaining a clean label is difficult. There is also a major need for products that prevent sarcopenia. GLP-1 users often lose muscle mass along with fat. This creates a significant opportunity for functional foods that deliver high-quality, bioavailable protein in small portions.
Functional foods now serve two related but different jobs. One is to stimulate the body’s own GLP-1 response in people not taking drugs. The other is to support people already on GLP-1 therapies, where the problem shifts to nutrient density, muscle retention, digestive tolerance, and hydration. That second use case is driving rapid product launches, but it should not be confused with proven food-based enhancement of endogenous GLP-1 physiology.
Current solutions also struggle with shelf life and cost. Nutrient-dense foods are often more expensive to produce than standard processed goods. In 2026, the industry is searching for ways to make these functional benefits accessible to a wider audience. We are seeing a shift from “diet” branding to “metabolic health” branding. This reflects a more scientific approach to product development.
Research Activity Overview
Research volume has accelerated sharply since 2020, with over 40 peer-reviewed publications on endogenous GLP-1 stimulation through food-based interventions and multiple active patent filings across US, KR, CN, and JP jurisdictions.
The field has also shifted from broad ingredient classes to mechanism-led systems. Earlier work often framed fiber or protein as general satiety tools. Newer work asks which receptors are activated, which microbial metabolites form, and whether nutrients reach distal L-cells intact.
Academic research dominates, but the industry share is increasing. Universities continue to publish on receptor pathways in enteroendocrine L-cells, short-chain fatty acids, ileal brake physiology, and food-derived peptides.
Industry activity is more translational. Companies are showing up through product-linked trials and platform launches. They are using known ingredients, such as beta-glucan, resistant starch, protein systems, probiotics, and postbiotics to build products for GLP-1 users. Olipop-backed work on prebiotic sodas, plant-extract trials measuring incretin response, and corporate R&D programs from Nestlé and Danone all point to an active translational push.
Academic-industry boundaries are blurring through strategic partnerships. Nestlé is working with UC Davis and Australian Catholic University to develop new ideas for weight management innovation incubators. At the same time, Meiji is filing patents on fermented milk-based GLP-1 secretagogues, based on academic research into how short-chain fatty acids affect gut cells.
Recent data shows that patent filings for GLP-1 stimulating ingredients have doubled in the last two years. Most of these patents involve fiber blends and plant extracts. Biotech companies are now partnering with major food manufacturers to bring specialized ingredients to market.
We are also seeing more research into the microbiome. Scientists are finding that specific gut bacteria play a huge role in how much GLP-1 the body produces. This has led to a surge in probiotic and prebiotic research.
The strongest shift is toward biological systems rather than single magic ingredients. That means the field is moving from “which ingredient boosts GLP-1” to “which food system creates a reliable postprandial signal with acceptable tolerance.” This is a more mature research question, and it aligns better with real product development.
GLP-1 Technologies Powering Innovation in Functional Foods
1. Fiber-maxxing and short-chain fatty acids to stimulate GLP-1
While fiber is known to help with weight management, the industry is struggling to identify which specific fibers most effectively convert into short-chain fatty acids that trigger GLP-1.
Instead of “bulk” fiber, research is accelerating in Propionate-producing fibers. By engineering specialized prebiotic blends (acetylated starches), food scientists can ensure that the gut microbiome produces high levels of Propionate and Butyrate, which are the primary chemical signals the gut uses to release GLP-1.
These acids bind to receptors on L-cells and trigger hormone release. R&D teams are addressing the “grittiness” problem in high-fiber products. New processing techniques enable high-fiber loads in beverages and snacks without compromising texture. This technology is critical because it offers a natural way to boost satiety.
2. Countering rapid enzymatic degradation of incretins
Natural GLP-1 produced by the body has an incredibly short half-life of about 2 minutes because the enzyme DPP-4 breaks it down almost instantly.
This rapid clearance means that even if a food triggers a spike in GLP-1, the metabolic “fullness signal” vanishes before it can effectively suppress appetite or regulate glucose over a sustained period.
R&D is shifting toward identifying specific peptides derived from whey, soy, and amaranth that act as natural DPP-4 inhibitors. By incorporating these peptides into functional foods, the “cleanup crew” enzymes are occupied, allowing the body’s natural GLP-1 to circulate and signal fullness for longer.
Key innovation includes the use of In-silico Molecular Docking to predict which plant-based peptides will bind most tightly to the DPP-4 active site, moving beyond trial-and-error kitchen science.
3. Resolving the sensory-efficacy conflict of bitter agonists
Recent research has revealed that bitter taste receptors (T2Rs) are not just on the tongue; they are also located on L-cells in the gut. Activating these gut receptors is one of the fastest ways to increase GLP-1 levels in the system. However, consumers generally reject highly bitter foods.
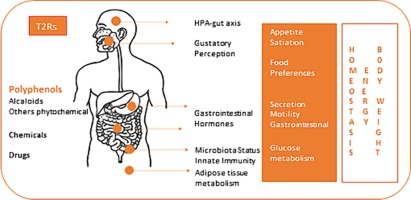
Research involves microencapsulating potent bitter compounds that remain tasteless in the mouth but “bloom” or activate once they reach the stomach and intestines, triggering the GLP-1 response without compromising the food’s flavor profile.
With aging, TAS2R gene activity declines. However, grape-seed extract (GSPE) can help restore their expression. These receptors also act as key connectors between gut lining health, inflammation, and beneficial bacteria that produce butyrate.
4. Bypassing synthetic dependence with plant-based mimicry
Synthetic GLP-1 agonists are highly effective but come with high costs and potential gastrointestinal side effects. There is a massive market gap for “Nature’s Agonists” food-derived compounds that can stimulate the GLP-1 receptor directly or enhance its sensitivity without being a pharmaceutical drug.
This area leverages AI to screen thousands of botanical compounds for GLP-1 secretagogues. Key molecules under investigation include berberine, curcuminoids, and ginsenosides. These compounds work by activating the AMPK pathway, which indirectly boosts GLP-1 secretion and improves insulin sensitivity.
High-throughput screening of traditional medicinal herbs to find “small molecule” plant compounds that mimic the docking behavior of pharmaceutical GLP-1 drugs, allowing for the creation of “Medical Grade” functional foods.
5. Lean body mass loss risk in weight management
Maintaining muscle is a top priority for GLP-1 users. Lean body mass loss risk occurs during the administration of GLP-1 receptor agonists and metabolic therapies. Mitigating this loss protects the long-term commercial viability and safety margin of metabolic therapies.
Research focuses on “fast” and “slow” protein blends to maximize muscle protein synthesis. Scientists are using enzyme-assisted systems to improve the body’s absorption of these proteins. This is especially important for users who are eating much smaller portions. The goal is to provide 20 to 30 grams of high-quality protein in a very small volume of food.
Coordinating multiple agonist activities addresses the ceiling effect of monotherapy on glucose regulation and adiposity.
Startup and Corporate Innovation Activity
The startup landscape is splitting into two main groups. One group focuses on simple, low-cost approaches, such as using existing fibers and proteins for GLP-1 marketing positioning. The other group invests heavily in advanced technologies like AI-designed peptides and microbiome-based solutions.
Corporate food manufacturers are adopting portfolio relabeling strategies that require minimal R&D investment but offer limited defensibility. The real progress will depend on strong clinical validation at the supplement level investment to stand out beyond basic marketing claims.
Emerging startups and technology platforms:
AI-Driven Peptide Discovery
- Lembas (Israel/UK) raised $3.6M to develop GLP-1 Edge™, in early funding led by FLORA Ventures. It is developing a GLP-1–related peptide using an AI platform from Tel Aviv University, designed for use in shakes and snacks.
- NotCo shifted focus to health products with “GLP Booster,” an AI-designed plant-based ingredient. Now supplies ingredients to food companies.
Clinically Tested Plant Extracts
- Calocurb (New Zealand) developed a hops extract (Amarasate®) after 14 years of research. Three clinical trials demonstrate 30% reduction in hunger, 40% reduction in cravings, and 18% reduction in calorie intake within one hour.
- Appethyl, a spinach-based extract with positive clinical results showing 43% greater weight loss over 12 weeks and up to 95% reduction in hedonic hunger. It faced setbacks after EFSA’s 2023 rejection of weight management health claim. It has since been relaunched.
Microbiome and Fiber-Based Solutions
- Supergut has raised over $100M expansion to 6,000+ retail doors. GLP-1 Booster delivers 6g patented prebiotic fiber blend per serving. A peer-reviewed 12-week clinical study demonstrated improved glycemic control in type 2 diabetics.
- One Bio attracted $27 million to commercialize short-chain fiber ingredients that can be incorporated into plant milks, cereals, and sparkling water without causing sensory degradation.
- Pendulum Therapeutics developed GLP-1 Probiotic by combining Akkermansia muciniphila, Clostridium butyricum, and Bifidobacterium infantis. Clinical data indicate 91% of users report reduced food cravings after six weeks.
- Akarso Bio developed “uniquely structured prebiotic fiber” claiming GLP-1 stimulation without gastrointestinal side effects, with stated IP portfolio.
- Olipop is tied to a current pilot study on the effects of prebiotic soda on GLP-1, PYY, transit time, and satiety.
Large food companies:
- Nestlé has launched a brand called Vital Pursuit. This line features portion-controlled meals designed specifically for GLP-1 users. It then expanded its weight management platform with whey protein microgel technology aimed at appetite support, blood sugar management, and muscle preservation in beverage form.
- Conagra has labeled 24+ existing Healthy Choice meals meeting low calorie, high protein, and fiber thresholds as “GLP-1 friendly.”
- Medifast launched Optavia Ascend, a line of high-protein, fiber-rich mini meals and nutrient packs positioned for people using GLP-1 drugs.
- Smoothie King launched a GLP-1 Support Menu built around high-protein, no-added-sugar smoothies.
- General Mills positioned GLP-1 trends as a snack opportunity, innovating with Cheerios Protein and Nature Valley protein bars. They are targeting GLP-1 users with advertising for high-protein, high-fiber soups.
- Danone followed with Oikos Fusion, a cultured dairy drink built around whey, leucine, and vitamin D for muscle support and digestive health during weight loss, including for GLP-1 users.
Ingredient suppliers building the B2B layer:
- ADM launched a platform for GLP-1 and anti-obesity medication users built around formulation support and its ingredient portfolio.
- Glanbia has framed weight management in the GLP-1 era as a nutrition design problem, with an emphasis on protein, hydration, digestive health, and reduced sugar intake.
- Kerry is positioning postbiotics and probiotics around digestive comfort and quality of life for GLP-1 users, which is a smart move because gastrointestinal side effects are one of the main reasons people stop treatment.
Partnership strategies and value chain evolution:
- Shiru’s GLP-1 Innovation Alliance unites Fortune 500 CPG companies and emerging brands to democratize AI-powered ingredient discovery through the Flourish™ platform. It contains 33+ million proteins and operates tiered business models that challenge proprietary R&D exclusivity.
- Nestlé is working with universities such as UC Davis and Australian Catholic University to develop science-based solutions for weight management.
- Morrisons partnered with Applied Nutrition in a 3-year deal to launch 53 high-protein products across UK stores.
- Co-op and M&S introduced smaller meals and nutrient-rich salads to capitalize on GLP-1 trends.
Regulatory and Compliance Landscape
United States: Structure function boundary enforcement
FDA allows structure and function claims for conventional foods, but the line between those claims and disease claims still matters. A food can claim to support normal body functions if it is truthful and not misleading. It cannot position itself as treating obesity or functioning like a drug without stronger regulatory backing.
The FDA has taken a strict approach to enforcing regulations in this space.
In December 2024, it issued warning letters to companies like Prime Peptides, Summit Research Peptides, Swisschems, and Xcel Peptides for promoting unapproved GLP-1 receptor agonists labeled as “for research use only.”
Later, in September 2025, a warning letter to GLP-1 Solution highlighted that compounded retatrutide, semaglutide, and tirzepatide were being sold as unapproved and misbranded drugs.
And in early 2026, the FDA issued 30 warning letters to companies making misleading GLP-1 claims. Most of these were telehealth firms, but the message was clear for food companies too. You cannot claim a food is a “natural Ozempic” without clinical proof.
The FDA states that any product that claims to treat diabetes or obesity, or suggests similarity to approved GLP-1 drugs, is considered an unapproved drug under federal law. Additionally, Import Alert 66-80 blocks low-quality GLP-1 ingredients from entering the U.S.
European Union: Health claim restrictions on GLP-1
In the EU, the claims framework is tighter because health claims must be listed in the EU Register and meet its conditions of use. The register includes both authorised and non-authorised claims. That means companies need to check whether a satiety or metabolic claim route is already recognised before they move it into packaging or advertising.
There is a push for more transparency in how ingredients are sourced. Novel food applications for GLP-1 boosters are under heavy scrutiny. This makes it harder for companies to use completely new ingredients. Most are sticking to GRAS materials but using them in new ways.
The EFSA has not approved any health claims that directly refer to GLP-1 hormone modulation. Products must instead focus on approved markers backed by well-understood mechanisms.
South Korea (MFDS): Acceptance of GLP-1 as a biomarker
In January 2025, Korea Ginseng Corporation received MFDS approval for red ginseng to support blood sugar control. This approval was based on a 12-week clinical study in prediabetic adults, which showed a 9.9% increase in GLP-1 levels.
This decision is important because MFDS accepted GLP-1 as a valid biomarker, unlike EFSA’s more cautious approach.
From June 2025, MFDS has also introduced a faster regulatory process, recognizing global safety data and simplifying updates that do not affect product safety.
Japan: No clear pathway for GLP-1 claims
Japan’s FOSHU system, which had around 11,000–12,000 approved products by FY2023, does not provide a clear route for GLP-1-related claims.
Most approved products focus on digestive health, especially through dietary fiber. Claims related to blood sugar control, such as those using L-arabinose or white sweet potato peptides work through mechanisms that do not involve GLP-1.
An alternative route is the Functional Labeling Foods (FFC) category. However, only about one-third of approved products succeed in the market due to strong competition. This pathway also avoids direct references to GLP-1.
Grey zones and emerging scrutiny
“GLP-1 friendly” as implied disease claim: The terms “GLP-1 friendly,” “GLP-1 support,” and “natural GLP-1 booster” can be risky. Phrases like these may be interpreted as implying obesity treatment, which could trigger FDA enforcement. Regulators are likely to crack down on these labels if they imply medical equivalence. R&D teams must work closely with legal departments to ensure that claims are supported by peer-reviewed studies.
Prebiotic/probiotic GLP-1 claims: While the microbiome may influence GLP-1 through short-chain fatty acids (SCFAs), regulators have not yet defined whether this counts as a general health claim or a disease-related claim.
Cross-border e-commerce: Agencies like the FDA and Australia’s TGA are focusing on direct-to-consumer sales of unapproved GLP-1 products, and foods making strong GLP-1 claims may face heightened scrutiny.
No major regulatory movement specific to GLP-1 foods has emerged beyond broader claim enforcement and existing food labeling rules. The likely next phase is tighter scrutiny of wording, substantiation quality, and the boundary between companion nutrition for drug users and implied pharmacological action.
Key Bottlenecks and Unresolved Challenges in GLP-1 Functional Foods
The biggest unresolved issue is the human dose response. Many ingredients can influence GLP-1 signaling in vitro or in tightly controlled settings, but human responses in finished foods are smaller and less consistent. The response depends on baseline metabolic health, microbiome composition, meal context, and dose. That makes claim support hard and product performance uneven.
Sensory impact is another major hurdle. Masking the flavor of potent fibers and bitter compounds is difficult. Many current products still have an “aftertaste” that consumers dislike. Fiber-rich systems can create viscosity and digestive discomfort. Bitter agonists face taste barriers. Protein-fortified small meals often become chalky or dense. If the consumer is already dealing with nausea or slowed gastric emptying from GLP-1 medication, a poorly designed food can increase rejection.
Bioavailability of food-based GLP-1 boosters. While a drug can provide a massive dose of the hormone, food triggers a much smaller response. It has not been proven that these small increases are sufficient to cause significant weight loss on their own. Long-term studies are needed to determine whether the body develops a tolerance to these natural boosters. A main remaining challenge is that they are stable under laboratory conditions but often fail to remain stable during real industrial processing.
Scale-up reliability is still fragile. R&D can make a high-fiber, high-protein, microbiome-active prototype in the lab. Scaling that system into an affordable shelf-stable product with stable texture, clean labeling, and broad sensory acceptance is harder. This is where many promising concepts will stall.
Bioavailability and delivery system cost burden. Native GLP-1 has a very short half-life of just 1–2 minutes due to rapid degradation by DPP-4 and renal clearance.
Patents have been filed in pH-responsive microspheres, nanoliposomes with modified surfaces, and self-assembling peptide hydrogels. However, most of these solutions are still at the pilot stage and not widely commercialized. The major issue is cost here. Advanced manufacturing methods make large-scale production expensive.
Formulation trade-off. Fermentable fibers can help GLP-1 pathways, but they can also raise bloating risk. Protein hydrolysates can be active, but they can also create bitterness and stability problems. Live biotics face shelf life and viability constraints. Distal delivery systems add cost and process complexity.
White Space and Strategic Opportunities in GLP-1 Functional Foods
Pediatric and adolescent metabolic health
A carbohydrate blend of raw corn starch and raw potato starch has been shown to stimulate GLP-1 release in obese children. A two-month study reported reduced appetite and food intake. This highlights food-based solutions as a practical alternative for families reluctant to use drugs.
Rising childhood obesity globally creates strong opportunities in this segment. Unlike adult markets where pharmaceutical alternatives dominate, pediatric applications face higher regulatory scrutiny for drug interventions, creating favorable conditions for food-based approaches.
Veterinary and pet food applications
GLP-1-based solutions are emerging in pet care. Cornell University’s trials in cats show weight loss benefits from long-acting therapies, while supplements targeting GLP-1 pathways are entering the dog market. The veterinary market offers potential for technology transfer to human applications. Pet food industry can serve as a testing ground for new delivery systems and ingredients before humans.
Sports nutrition and muscle preservation
15–60% of weight loss from GLP-1 drugs comes from lean muscle mass, with potential losses of 7.5–30 pounds of muscle when shedding 50 pounds.
There is a substantial opportunity for “GLP-1 companion” sports nutrition products that support muscle retention, such as high-protein, nutrient-dense formats specifically formulated to preserve strength during weight loss.
Current products such as Nestlé Boost (35g protein, fiber, and micronutrients) and Remedy Health’s Collagen+ Gut gummies address nutritional gaps. But targeted sports nutrition formulations that combine support for protein synthesis with GLP-1-friendly digestive profiles remain underdeveloped.
Savory, shelf-stable, nutrient-dense formats
Most visible launches lean on frozen meals, shakes, and powders. There is room for clinically informed soups, savory snacks, bakery formats, and small meals that offer better satiety and easier tolerance. The science on food structure and gastric emptying supports this path, but commercial work is still in its early stages.
Cross-industry transfer from pharma delivery into food structure design
Delayed-release, targeted intestinal delivery, and receptor-specific trigger combinations are common concepts in drug science. Food systems have only recently begun to apply them to support satiety and glycemic control. Companies that can bring this precision into everyday formats will have an edge.
Food-grade alternatives using alginate-chitosan systems (CN116421579A) show promise but require further development to match pharmaceutical performance.
Elderly nutrition market
Sarcopenia or muscle loss is a major problem for the aging population. Functional foods designed for GLP-1 users can be easily adapted for this group. Both groups need high-protein, nutrient-dense, and easy-to-digest options. This cross-industry potential is often overlooked.
Personalized nutrition
In 2026, more people are using DNA and blood tests to guide their diet. Creating “GLP-1 kits” that are tailored to an individual’s gut microbiome is a high-potential area. This moves the industry away from a “one size fits all” approach.
Geographic market opportunities
China is projected to become the world’s leading GLP-1 market by 2030, reaching RMB 100 billion (US$14 billion), with ~200–250 million obese adults anticipated. ~50% of adults and 20% of children in China are obese, driven by rising obesity rates and Western-style dietary shifts.
High drug costs and limited insurance coverage are driving demand for affordable, food-based alternatives, supported by strong regional innovation in functional foods.
APAC heart-health functional food market demonstrates regional innovation capacity, with omega-3 fatty acids, plant sterols, soluble fibers, and probiotics central to formulations in China, Japan, India, and Australia. GLP-1-relevant extensions of these established ingredient platforms represent near-term commercialization opportunities.
What to Monitor Next
Watch ingredient systems that combine fermentable fiber, protein quality, and meal structure. That is where the strongest science and the lowest regulatory risk meet.
Track company-led trials with finished foods, not just ingredient dossiers. The field needs more data on repeat use, tolerance, and actual meal-replacement behavior. Products designed for GLP-1 users will continue to launch, but only a small share will demonstrate durable adherence and measurable nutritional benefits.
Invest in technologies that improve the sensory profile of functional ingredients. The winner in this space will be the company that can deliver drug-like benefits with the taste and texture of a premium snack. Pay attention to startups working on encapsulation and flavor masking.
The challenge for R&D teams is not a lack of information. It is the fragmentation of signals across research papers, patents, startup activity, and early product launches. Teams struggle to connect ingredient-level insights to real product formats, assess which technologies are ready for scale, and identify which claims will hold under regulatory scrutiny. Timing also remains a problem, since by the time market trends become visible, the early advantage is already lost.
Slate, an R&D intelligence tool for GLP-1 addresses these gaps by bringing together research, patent activity, startup breakthroughs, market signals, and product innovation into a single view. It helps teams identify which ingredient systems are gaining traction, track how companies are translating science into products, and evaluate where white spaces exist. It allows R&D teams to move from scattered insights to clear decisions on what to build, partner, or prioritize before the market matures.


