If your cosmetic products contain perborates, silver compounds, or hexyl salicylate above new concentration limits, you have less than 90 days to pull them from EU shelves. Omnibus VIII isn’t giving you the courtesy of an adaptation period or grace window for existing inventory. May 1, 2026, is the hard stop, and every product sitting in your warehouse or on retail shelves after that date is non-compliant.
Here’s what you need to know, which ingredients are affected and what you should be doing now to stay ahead of enforcement risk.
Companies and Product Categories Most Affected
Omnibus VIII will have a widespread impact across the cosmetics industry, particularly affecting manufacturers with extensive portfolios in these categories:
- Fragrance houses and perfume manufacturers: Due to hexyl salicylate restrictions across hydroalcoholic fragrances, fine perfumes, and scented leave-on products
- Oral care brands: Restrictions on silver compounds and hexyl salicylate in toothpaste and mouthwash formulations require immediate reformulation
- Children’s cosmetics producers: Blanket prohibition of hexyl salicylate in products for children under 3 years old
- Color cosmetics manufacturers: Particle-size-dependent restrictions on CI 77820 (silver powder) in lip products and eyeshadows
- Preservative-dependent formulations: Brands using o-phenylphenol or sodium o-phenylphenate in leave-on and rinse-off products need concentration adjustments
- Natural and “clean beauty” brands using silver: Nano and non-micronised silver used as preservatives or antimicrobial agents are now completely banned.
Banned Ingredients Under Omnibus VIII
The regulation introduces several new prohibitions in Annex II (prohibited substances) that affect preservatives, oxidizing agents, technical ingredients, and nanomaterials:
- Perboric acid and its salts (including perborates): Previously regulated individually, now unified under a single ban due to concerns over hydrogen peroxide release and oxidative safety risks
- Solid silver (particle diameter ≥ 1 mm): Explicitly prohibited across all cosmetic applications
- Silver nanoparticles (1 nm < particle diameter ≤ 100 nm): Clear prohibition as a nanomaterial due to reproductive toxicity concerns and systemic accumulation risks
- N,N’-methylenediacrylamide: New CMR substance, fully prohibited across all cosmetic formulations regardless of concentration
- Trimethyl borate: New CMR substance banned from all cosmetic use
- Carbon nanotubes (various specific types): CMR classified and forbidden
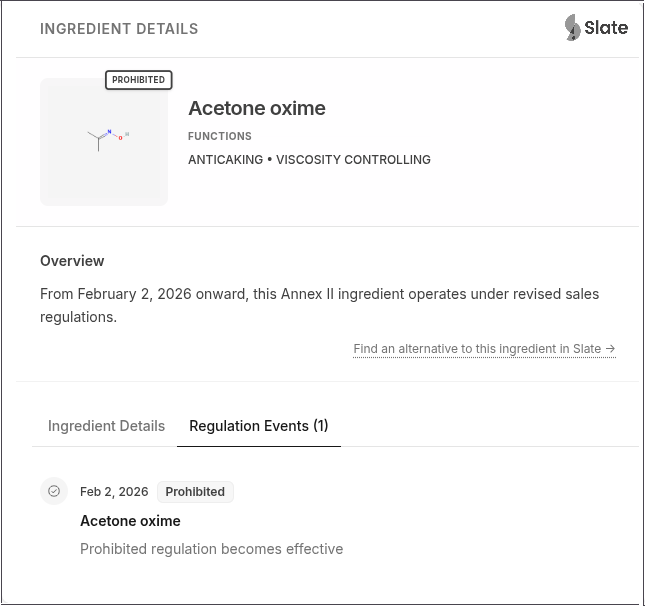
- Acetone oxime: New inclusion by CMR classification
New Restrictions on Key Ingredients
Beyond outright bans, Omnibus VIII introduces granular use restrictions across Annexes III, IV, and V that vary by product type, exposure route, and target population:
Annex III: Restricted Substances
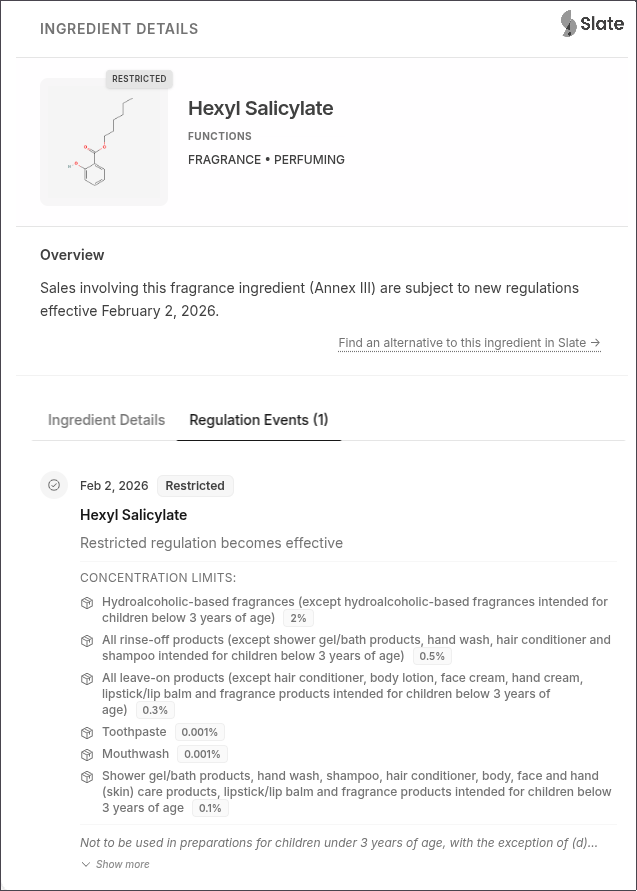
Hexyl Salicylate (CAS 6259-76-3): This fragrance ingredient now has highly differentiated concentration limits:
- Hydroalcoholic-based fragrances (except for children <3): 2% maximum
- Rinse-off products (except shower gel/bath, hand wash, hair conditioner, and shampoo for children <3): 0.5% maximum
- Leave-on products (except hair conditioner, body lotion, face cream, hand cream, lipstick/lip balm, and fragrance products for children <3): 0.3% maximum
- Toothpaste: 0.001% maximum
- Mouthwash: 0.001% maximum
- Shower gel/bath products, hand wash, shampoo, hair conditioner, body/face/hand care, lipstick/lip balm, and fragrance products for children <3: 0.1% maximum
- Note: Not permitted in preparations for children under 3 years of age, with the exception of toothpaste (0.001%) and the 0.1% category above
Silver (powder form) [100 nm < particle diameter ≤ 1 mm]: Permitted only in toothpaste and mouthwash at a maximum concentration of 0.05% in the final cosmetic product.
Annex IV: Allowed Colorants
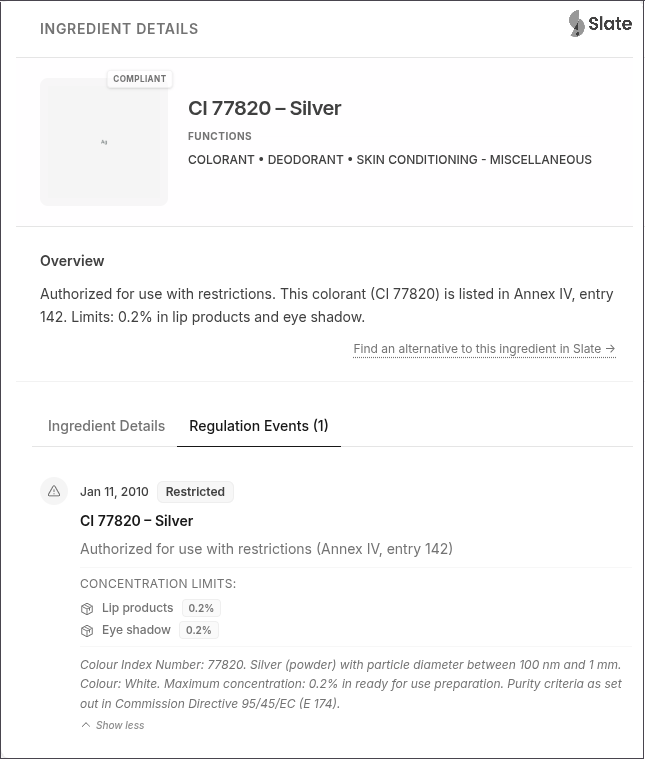
CI 77820 – Silver (powder form) [100 nm < particle diameter ≤ 1 mm]: Permitted only in lip products and eye shadow at a maximum concentration of 0.2% in the final cosmetic product.
Annex V: Allowed Preservatives
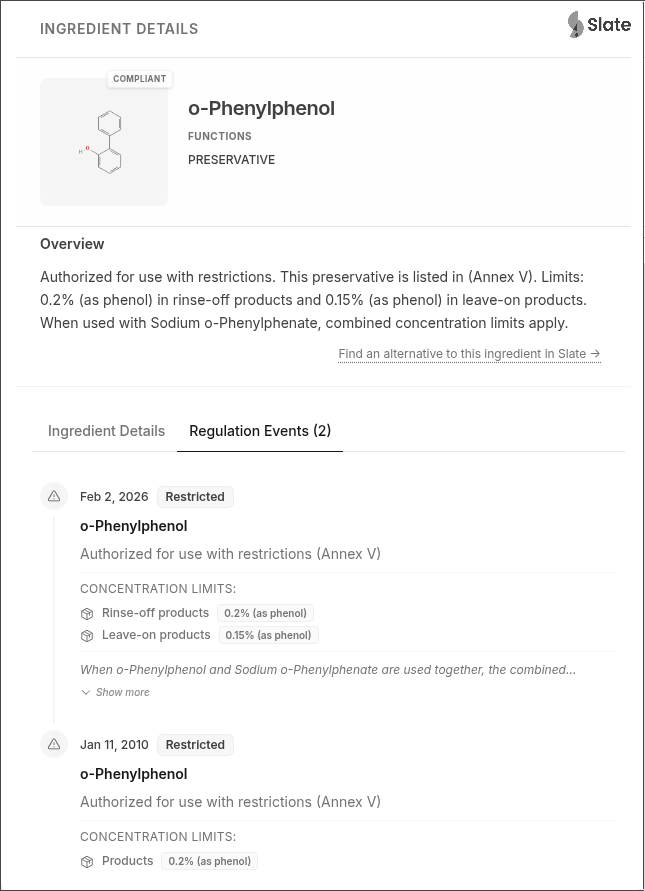
o-Phenylphenol and Sodium o-Phenylphenate: Updated preservative restrictions:
- Rinse-off products: 0.2% maximum (as phenol)
- Leave-on products: 0.15% maximum (as phenol)
- When used together, combined concentration (as phenol) shall not exceed 0.2% in rinse-off and 0.15% in leave-on products
- Not to be used in applications that may lead to exposure of the end-user’s lungs by inhalation
- Not to be used in oral products
- Mandatory label warning: “Avoid contact with eyes”
Summary of Ingredient Bans and Restrictions Under Omnibus VIII
| Ingredient / Substance | Regulatory Status Under Omnibus VIII | Annex | Key Conditions / Notes | Product Categories Impacted |
| Perboric acid and its salts (perborates) | Prohibited | Annex II | Unified ban due to CMR concerns and hydrogen peroxide release | Hair bleaching, oral care, whitening formulations |
| Silver nanoparticles (1–100 nm) | Prohibited (Nanomaterial) | Annex II | Complete ban due to reproductive toxicity and systemic exposure risks | Antimicrobial, “clean beauty,” preservative systems |
| Solid silver (≥ 1 mm particle size) | Prohibited | Annex II | Explicitly excluded from cosmetic use | Specialty and decorative formulations |
| Silver powder (100 nm – 1 mm) | Restricted use only | Annex III / IV | Allowed only in specific product types and concentrations | Toothpaste, mouthwash, lip products, eye shadow |
| Hexyl Salicylate (CAS 6259-76-3) | Restricted (category-specific limits) | Annex III | Multiple concentration limits by product type; generally not permitted for children <3 (with narrow exceptions) | Fragrance, leave-on, rinse-off, oral care |
| N,N′-Methylenediacrylamide | Prohibited (CMR) | Annex II | Newly classified CMR substance; no permitted cosmetic use | Polymer-based and technical formulations |
| Acetone oxime | Prohibited (CMR) | Annex II | Added following CLP classification update | Industrial/technical ingredient users |
| Trimethyl borate | Prohibited (CMR) | Annex II | No concentration exemptions permitted | Specialty and functional formulations |
| Carbon nanotubes (specified types) | Prohibited (CMR) | Annex II | Applies to listed CNT variants with CMR classification | Advanced / functional cosmetic technologies |
| o-Phenylphenol & Sodium o-Phenylphenate | Restricted preservative | Annex V | Max 0.2% (rinse-off) / 0.15% (leave-on); inhalation and oral use prohibited; label warning required | Preservative-dependent formulations |
| UV filters | No change under Omnibus VIII | Annex VI | Not affected by this regulation | Sunscreens, SPF cosmetics |
Major multinational cosmetics companies like L’Oréal, Unilever, Estée Lauder, Procter & Gamble, and their subsidiaries will need to conduct comprehensive ingredient audits across thousands of SKUs to identify at-risk formulations.
Navigate Compliance with Slate: Your Risk Intelligence Platform
Managing regulatory compliance across a global product portfolio is complex, especially when deadlines are tight and the stakes are high. Slate provides cosmetic brands with an advanced ingredient intelligence database that helps you proactively identify regulatory risks before they become compliance crises.
Instead of manually cross-referencing thousands of formulations against evolving annexes, Slate automates risk detection and empowers your R&D and regulatory teams to make data-driven reformulation decisions.
Whether you’re discovering alternative ingredients, managing reformulation timelines, conducting supplier audits, or preparing for market surveillance inspections, Slate gives you the visibility and control you need to stay ahead of regulatory change.
Don’t let May 1, 2026, catch you unprepared. Discover which of your products contain Omnibus VIII-restricted ingredients and take action today.


